#Cyclic Amine
Explore tagged Tumblr posts
Text
Polyethyleneimine: An Important Building Block in Various Applications In Industry

What is Polyethyleneimine?
Polyethyleneimine (PEI) is a polymeric compound composed of repeating units of ethyleneimine monomers. It is a white solid polymer that is soluble in water. PEI is classified as a branched or linear cationic polymer that is formulated through the polymerization of aziridine. The polymer's molecular weight can range from 300 to over 10,000 Daltons depending on its intended use. Properties and Structure of PEI
PEI is a polymer that contains secondary nitrogen atoms along its backbone that provide cationic charge centers. These nitrogen atoms can interact and form electrostatic bonds with negatively charged macromolecules or surfaces. The cationic nature of PEI allows it to act as a polycation and complex or condense with negatively charged substances like DNA or RNA. PEI is also very hygroscopic due to its numerous amine groups that attract water molecules. Its branched structure provides greater buffering capacity and DNA condensation ability compared to the linear form of PEI. Uses in Gene Delivery
One of the most important uses of Polyethyleneimine is in non-viral gene delivery applications. Its cationic charge allows it to efficiently bind and compact negatively charged nucleic acids like plasmids, forming nano-sized polyplexes that can transfect cells. The polyplexes are effectively endocytosed by cells and the low pH of endosomes triggers PEI to buffer the compartment, releasing the DNA from the complex. This "proton sponge" mechanism of PEI facilitates effective intracellular delivery and expression of the gene of interest. It is commonly used as a transfection agent for in vitro studies as well as in vivo gene therapies and vaccines. Role in Biomaterials and Tissue Engineering
Given its favorable properties, PEI also finds extensive use as biomaterials for tissue engineering applications. Its cationic nature permits binding of PEI to negatively charged components of the extracellular matrix like glycosaminoglycans. This helps direct cell adhesion, growth, and function. PEI can act as a coating onto implants and scaffolds, promoting their integration with surrounding tissues. It allows bioconjugation of signaling cues to influence cell behavior in regenerative therapies as well. The tunable properties of PEI through controlling its molecular weight or branching also expand its utility in the design of biomaterials.
In addition to the major uses mentioned above, PEI has applications in many other fields due to its versatile characteristics. It acts as a flocculating agent in wastewater treatment processes to remove pollutants and purify water. PEI finds increasing importance as a hardener in epoxy resins used in manufacturing infrastructure and electronics. Its buffering ability makes it valuable as a non-corrosive alternative to replace hazardous chemicals in industrial cleaning agents as well. Overall, the unique polymeric structure and properties of PEI designate it as an essential building block material with myriad applications.
Get more insights on Polyethyleneimine
Unlock More Insights—Explore the Report in the Language You Prefer
French
German
Italian
Russian
Japanese
Chinese
Korean
Portuguese
Alice Mutum is a seasoned senior content editor at Coherent Market Insights, leveraging extensive expertise gained from her previous role as a content writer. With seven years in content development, Alice masterfully employs SEO best practices and cutting-edge digital marketing strategies to craft high-ranking, impactful content. As an editor, she meticulously ensures flawless grammar and punctuation, precise data accuracy, and perfect alignment with audience needs in every research report. Alice's dedication to excellence and her strategic approach to content make her an invaluable asset in the world of market insights.
(LinkedIn: www.linkedin.com/in/alice-mutum-3b247b137 )
#Polyethyleneimine#Aliphatic Carbon#Versatile Polymer#PEI#Linear Polymer#Cyclic Amine#Cationic Polymer#Biopolymer#Flocculant#Emulsifier#Nanoparticle Synthesis
0 notes
Text

Innovative porous organic crystal structure offers superior CO₂ separation
Porous organic crystals with superior properties as CO2 adsorbents were created by researchers at Institute of Science Tokyo. Owing to the novel 2.5-dimensional skeleton, the materials feature ultrahigh-density amines. The covalently-bonded microporous skeleton and high crystallinity realize fast CO2 adsorption and high thermal stability. Their low adsorption heat, only one-fourth of the current amine scrubbing method, and their light-elemental nature can reduce the cost for CO2 separation from flue gases. To mitigate climate change, CO2 emissions from large industrial facilities have to be reduced. To separate CO2 from the flue gases, the major current technology is the amine scrubbing method, in which an aqueous solution of amine molecules is circulated in the capture facility to cyclically perform capture and release of CO2. However, this method suffers from reportedly high running costs and several problematic aspects of amine solutions such as high environmental risk and corrosivity to steels.
Read more.
10 notes
·
View notes
Text
CAS NO. 35180-01-9 Manufacture
Understanding CAS No. 35180-01-9: A Comprehensive Guide
CAS numbers are unique identifiers used to track chemical compounds with precision. They help researchers, manufacturers, and regulatory agencies identify substances across various industries and applications. CAS No. 35180-01-9 is tied to a compound called N-ethyl-2-pyrrolidone (NEP), an organic chemical compound that may not be as widely recognized as others but still plays a crucial role in several industries. This guide explores the nature, applications, and safety considerations of NEP, shedding light on its importance.

What Is N-Ethyl-2-Pyrrolidone (NEP)?
N-ethyl-2-pyrrolidone (NEP) is an organic solvent that belongs to a group of chemicals called pyrrolidones. These are characterized by a five-membered lactam ring structure, which is a cyclic amide. NEP is commonly used in various industries because of its ability to dissolve both hydrophobic (water-repelling) and hydrophilic (water-attracting) substances. This makes it highly versatile in a wide range of applications.
NEP is typically a colorless to pale yellow liquid with a slightly amine-like odor. Its molecular formula is C6H11NO, and it has a molecular weight of 113.16 g/mol. The compound has a boiling point of 204°C and a melting point of -24°C, which gives it a broad range of applications under different temperature conditions.
Key Chemical Properties of NEP
Appearance: Colorless to pale yellow liquid with a faint, amine-like smell.
Molecular Formula: C6H11NO.
Molecular Weight: 113.16 g/mol.
Boiling Point: 204°C.
Melting Point: -24°C.
Solubility: NEP is highly soluble in water and many organic solvents, which makes it a useful solvent for various chemical processes.
Major Applications of N-Ethyl-2-Pyrrolidone (NEP)
NEP is widely used in different industries due to its unique solvent properties, which allow it to dissolve a broad range of substances. Some of the primary applications of NEP are in chemical synthesis, pharmaceuticals, electronics, and agriculture.
1. Chemical Synthesis and Manufacturing
NEP plays an important role in the chemical synthesis of many products. It is often used as a solvent in chemical reactions, including polymerization (the process of creating polymers). Additionally, NEP is found in the production of paints, coatings, adhesives, and other chemical formulations. Its ability to dissolve both water-soluble and water-insoluble compounds makes it an excellent solvent in processes that require a combination of different chemicals. It aids in the creation of high-performance materials used in everything from coatings on cars to adhesives in electronics.
2. Pharmaceuticals and Biotechnology
In the pharmaceutical and biotechnology industries, NEP is used as a solvent to help dissolve active pharmaceutical ingredients (APIs) in the creation of medications. The solubility properties of NEP are critical to improving the stability of drugs and ensuring they can be delivered efficiently into the body. NEP helps improve the bioavailability of drugs, meaning it can enhance the absorption and distribution of medications in the bloodstream, leading to more effective treatments.
NEP is also used in drug delivery systems, such as injectable formulations and transdermal patches, where it ensures that the active ingredients remain stable and effective for a longer period. Its versatility is particularly useful when formulating medications that need to be absorbed through different methods, whether orally, intravenously, or through the skin.
3. Electronics and Battery Technology
In the electronics and battery industries, NEP is used in the formulation of electrolytes for lithium-ion batteries. Electrolytes are crucial for the functioning of rechargeable batteries, and NEP helps improve their ionic conductivity, which in turn enhances the performance and longevity of these batteries. Lithium-ion batteries are widely used in consumer electronics like smartphones, laptops, and electric vehicles. By improving the electrolyte's conductivity, NEP contributes to increasing the efficiency and lifespan of these energy storage devices.
4. Agriculture
NEP is also used in the agricultural sector, where it serves as a solvent in the production of agrochemicals such as pesticides, herbicides, and fungicides. NEP helps dissolve the active ingredients in these chemicals, ensuring that they are well-distributed and effective when applied. Its ability to enhance the solubility and stability of these substances ensures that they are effective at controlling pests and diseases while being easier to apply.
Health and Safety Considerations
Like many industrial chemicals, NEP requires careful handling to minimize risks. While it is not classified as highly hazardous, it is still important to follow safety protocols to avoid health issues, especially when handling large quantities or in poorly ventilated areas.
Skin and Eye Contact: Prolonged or repeated exposure to NEP can cause skin irritation, and care should be taken to avoid direct skin contact. If contact occurs, wash the affected area thoroughly with soap and water.
Inhalation: Inhaling vapors of NEP can cause respiratory irritation. Adequate ventilation should always be used, particularly in enclosed spaces, and personal protective equipment (PPE) such as gloves, goggles, and respiratory protection should be worn if necessary.
Regulatory Oversight: NEP is not classified as a carcinogen or mutagen. However, regulatory agencies such as the U.S. Environmental Protection Agency (EPA) and the European Chemicals Agency (ECHA) monitor its use to ensure that it meets safety standards and that any potential risks to workers or the environment are minimized.
Environmental Impact
One of the advantages of NEP is that it is biodegradable, meaning it can break down naturally over time in the environment. However, like any chemical compound, NEP should still be handled responsibly to reduce its environmental footprint. Proper waste disposal methods must be followed, and companies must ensure that any discharges of NEP are within permissible environmental limits.
The environmental impact of NEP is still being studied, and ongoing research aims to better understand its long-term effects. Nonetheless, due to its biodegradability, it is considered less harmful to the environment compared to many other solvents and chemicals.
Conclusion
N-ethyl-2-pyrrolidone (CAS No. 35180-01-9) is a versatile and important compound used across several industries, including chemicals, pharmaceuticals, electronics, and agriculture. Its solvent properties make it an essential ingredient in the production of many products, from medications to batteries. However, like all industrial chemicals, it must be handled with caution to protect both human health and the environment.
By understanding its properties and applications, industries can continue to use NEP effectively while minimizing risks associated with its handling and disposal. Proper safety protocols and environmental responsibility are key to ensuring that the benefits of NEP are maximized without causing harm to people or the planet.
URL: For more information, visit connex pharma : CAS NO. 35180-01-9
0 notes
Text
Biogas Treatment Solutions: Enhancing Efficiency and Sustainability

Introduction
Biogas is a renewable energy source produced from organic waste through anaerobic digestion. While biogas is an environmentally friendly alternative to fossil fuels, it requires treatment before use to remove impurities such as hydrogen sulfide (H₂S), carbon dioxide (CO₂), and moisture. Effective biogas treatment solutions improve energy efficiency, reduce equipment corrosion, and ensure compliance with environmental regulations.
Importance of Biogas Treatment
Raw biogas contains various contaminants that can impact its usability and efficiency. The primary reasons for treating biogas include:
Enhancing Calorific Value: Removing CO₂ increases the methane concentration, improving energy output.
Reducing Corrosion: Hydrogen sulfide removal prevents damage to engines, pipelines, and storage tanks.
Eliminating Moisture: Dehydration prevents pipeline blockage and improves combustion quality.
Ensuring Regulatory Compliance: Treated biogas meets environmental standards and grid-injection requirements.
Key Biogas Treatment Technologies
Several technologies are available to purify biogas, each suited to different scales and applications.
1. Hydrogen Sulfide (H₂S) Removal
H₂S is a toxic and corrosive gas that must be removed before biogas can be used or upgraded. Common H₂S removal methods include:
Iron Oxide-Based Adsorption: Using iron oxide pellets to react with H₂S, forming iron sulfide.
Biological Scrubbing: Utilizing bacteria to oxidize H₂S into elemental sulfur.
Chemical Scrubbing: Using alkaline solutions (e.g., sodium hydroxide) to absorb H₂S.
2. Carbon Dioxide (CO₂) Removal
CO₂ dilution lowers biogas energy content and must be removed for high-purity applications like biomethane production.
Water Scrubbing: CO₂ is absorbed in water under pressure and separated from methane.
Pressure Swing Adsorption (PSA): A cyclic process that uses adsorbent materials to separate CO₂ from biogas.
Membrane Separation: Utilizing selective membranes to filter out CO₂ while retaining methane.
3. Moisture Removal (Dehydration)
Moisture can cause operational issues in gas engines and distribution networks. Common dehydration methods include:
Condensation and Cooling: Lowering biogas temperature to condense water vapor.
Silica Gel or Molecular Sieves: Adsorbent materials that capture moisture.
Glycol-Based Dehydration: Using glycol solutions to absorb water.
Advanced Biogas Upgrading Technologies
Beyond basic treatment, advanced upgrading technologies enable biogas to meet stringent pipeline and vehicle fuel standards.
Cryogenic Separation: Freezing out impurities at ultra-low temperatures.
Amine Scrubbing: Chemical absorption of CO₂ using amine-based solvents.
Biological Methanation: Converting CO₂ into methane using microbes, enhancing biogas yield.
Applications of Treated Biogas
Once purified, biogas has multiple applications:
Electricity and Heat Generation: Fuel for cogeneration plants and boilers.
Vehicle Fuel (Biomethane): A sustainable alternative to compressed natural gas (CNG).
Injection into Natural Gas Grids: High-purity biomethane can be blended with conventional gas supplies.
Industrial Use: Feedstock for chemical production and fuel for industrial boilers.
Conclusion
Biogas treatment solutions play a critical role in maximizing the efficiency, sustainability, and commercial viability of biogas energy. By implementing the right purification technologies, industries and municipalities can enhance energy output, reduce emissions, and contribute to a greener future. Investing in biogas treatment not only improves operational efficiency but also supports global efforts toward renewable energy adoption.
Call to Action
Looking to optimize your biogas production? Contact us today to learn about the latest biogas treatment solutions tailored to your needs.
#biogas generator#biogas plant#biogas power generation#chp combined heat and power#chp plant#cogeneration#combined heat and power#Biogas Treatment Solutions
0 notes
Text
Clenbuterol for Sale: Unlocking Its Benefits, Dosage Tips, and Side Effects to Watch
In the fitness and bodybuilding world, Clenbuterol has gained a strong reputation for its potential to aid in fat loss and improve athletic performance. Originally developed as a bronchodilator to treat asthma, Clenbuterol has also been widely adopted as a powerful fat-burning supplement.

>>>>>>>>> Click Here To Buy (Clenbuterol ) Official Website <<<<<<<<<
Click Here To Buy:
______________________________________________________________
Though it is not approved for use in humans in some countries, including the United States, Clenbuterol is still widely sought after by athletes, bodybuilders, and fitness enthusiasts for its stimulating properties and effectiveness in enhancing body composition.
In this article, we’ll cover everything you need to know about Clenbuterol—from its benefits and dosage recommendations to potential side effects and safety precautions.
What is Clenbuterol?
Clenbuterol, commonly known as "Clen," is a sympathomimetic amine that belongs to a class of drugs known as beta-2 adrenergic agonists. It was initially developed for use as a bronchodilator in treating respiratory conditions, such as asthma, by relaxing and widening the airways. However, its potent fat-burning and muscle-preserving properties have led to its popularity among athletes and bodybuilders. Clenbuterol is not an anabolic steroid, but it has effects similar to those of stimulants like ephedrine, increasing metabolic rate and thermogenesis in the body.
Unlike anabolic steroids that build muscle by altering hormonal balance, Clenbuterol works by stimulating beta-2 receptors in the body. This results in an increase in core body temperature, which accelerates metabolic processes, leading to higher energy expenditure and enhanced fat burning. Clenbuterol’s muscle-sparing qualities also make it a favorite in cutting cycles, allowing users to retain lean muscle mass while shedding body fat.
How Does Clenbuterol Work?
Clenbuterol’s effects on fat loss and energy come from its ability to stimulate the central nervous system (CNS) and activate beta-2 receptors. When these receptors are activated, several processes are triggered, including:
Increased Thermogenesis: Clenbuterol increases body temperature by raising the basal metabolic rate (BMR). This means that the body burns more calories at rest, leading to greater fat loss.
Enhanced Lipolysis: Clenbuterol promotes lipolysis, the breakdown of stored fat, by increasing the production of cyclic adenosine monophosphate (cAMP). This hormone-sensitive lipase helps release fatty acids from fat cells, which the body can then burn for energy.
Muscle Preservation: One unique benefit of Clenbuterol is its muscle-sparing effect. Unlike traditional weight-loss drugs, Clen helps users retain muscle mass even while on a calorie-deficient diet, making it ideal for bodybuilders who want to cut fat without sacrificing muscle.
Improved Oxygen Flow: Since it was originally intended as a bronchodilator, Clenbuterol increases airflow, which can enhance aerobic capacity and endurance. This allows athletes to train harder and for longer periods.
Due to these mechanisms, Clenbuterol has gained popularity in fitness and bodybuilding circles as a tool for fat loss and cutting cycles.
Key Benefits of Clenbuterol
Effective Fat Loss Aid: Clenbuterol’s thermogenic and lipolytic effects make it highly effective for fat loss. Users often report rapid reductions in body fat, even with moderate doses.
Muscle Retention During Cuts: Many fat burners lead to muscle loss, but Clenbuterol is known for preserving lean muscle mass while stripping away body fat. This makes it an attractive option during cutting cycles.
Enhanced Workout Performance: Clenbuterol’s stimulant effects boost energy and endurance, helping users push through intense training sessions and improve cardiovascular performance.
Increased Metabolic Rate: By elevating the basal metabolic rate, Clenbuterol helps users burn more calories around the clock. This effect remains even during periods of rest, which is crucial for ongoing fat loss.
Appetite Suppression: Clenbuterol can reduce hunger, making it easier to stick to a calorie-restricted diet and manage cravings.
While these benefits make Clenbuterol an appealing option, it’s crucial to use it responsibly and follow proper dosing guidelines to minimize side effects.
Clenbuterol Dosage Guidelines
Clenbuterol dosing should be approached with caution, especially for first-time users. The dosage can vary based on factors such as experience, body weight, and tolerance level. Here’s a general guideline to get started with Clenbuterol:
Beginner Dosage
For beginners, it’s recommended to start with a low dose of 20 mcg per day to assess tolerance. If no adverse reactions occur, the dose can be gradually increased by 20 mcg every few days until reaching the desired level. Many beginners cap their dosage at around 60–80 mcg per day.
Intermediate Dosage
Intermediate users often start at 40 mcg per day and may increase their dosage every 2–3 days. They usually peak at around 80–100 mcg per day. This dosage range provides effective fat-burning benefits without overly increasing the risk of side effects.
Advanced Dosage
Advanced users, who have built a tolerance, sometimes start with doses of 80 mcg or higher. These users may peak at 120–140 mcg per day. However, it’s essential not to exceed 140 mcg daily, as higher doses can lead to severe side effects.
Cycling Clenbuterol
Due to its stimulating effects, the body can quickly build a tolerance to Clenbuterol, reducing its effectiveness over time. Common cycle methods include:
Two Weeks On, Two Weeks Off: This approach involves taking Clenbuterol for two weeks, then taking a two-week break. This cycle prevents the body from adapting to the drug.
Incremental Cycling: Users gradually increase the dose over a two-week period, then take a break before starting another cycle.
It’s crucial to follow these cycles to prevent tolerance buildup and maintain Clenbuterol’s effectiveness.
Clenbuterol Stacking Options
Clenbuterol is often stacked with other compounds to enhance its fat-burning effects. Some popular stacking options include:
Anavar: Clenbuterol and Anavar (an oral anabolic steroid) are commonly stacked during cutting cycles. Anavar aids in preserving lean muscle while Clenbuterol promotes fat loss, making it an effective combination.
T3 (Cytomel): T3, a thyroid hormone, is often stacked with Clenbuterol for aggressive fat loss. However, this combination is only recommended for advanced users, as it significantly increases metabolic rate and can lead to severe side effects if misused.
Winstrol: Winstrol is another popular cutting steroid that pairs well with Clenbuterol. This combination is ideal for those looking to achieve a lean, shredded look with enhanced muscle definition.
It’s essential to research each compound thoroughly and understand the risks before creating any stack.
Potential Side Effects of Clenbuterol
Despite its fat-burning benefits, Clenbuterol is not without side effects, and misuse can lead to severe health risks. Here are some common side effects associated with Clenbuterol:
1. Increased Heart Rate and Blood Pressure
Clenbuterol can stimulate the cardiovascular system, leading to an elevated heart rate and increased blood pressure. High doses or prolonged use can lead to palpitations and, in severe cases, heart hypertrophy (an enlargement of the heart muscle).
2. Tremors and Muscle Cramps
Due to its stimulating effects, Clenbuterol can cause muscle tremors and cramps, especially in the hands. This is a common side effect, particularly at higher doses. Potassium supplementation and staying hydrated can help reduce cramps.
3. Insomnia and Restlessness
Clenbuterol’s stimulant properties can interfere with sleep, leading to insomnia and restlessness. It’s best to take Clenbuterol earlier in the day to minimize sleep disturbances.
4. Nervousness and Anxiety
Many users report feeling jittery or anxious while taking Clenbuterol. Reducing the dose or taking breaks can help alleviate these symptoms.
5. Electrolyte Imbalance
Clenbuterol can deplete potassium and taurine levels in the body, potentially leading to an electrolyte imbalance. Supplementing with potassium and taurine may help offset this risk.
6. Potential for Cardiac Hypertrophy
One of the more serious side effects is cardiac hypertrophy, an enlargement of the heart that can occur with long-term or high-dose Clenbuterol use. This condition can be life-threatening and underscores the importance of following responsible dosing guidelines.
How to Use Clenbuterol Safely
If you decide to use Clenbuterol, follow these guidelines to reduce potential risks:
Start with a Low Dose: Begin with the lowest effective dose and increase gradually to assess tolerance.
Stay Hydrated and Supplement Wisely: Clenbuterol can deplete electrolytes, so it’s essential to stay hydrated and consider potassium and taurine supplements to prevent cramps and imbalances.
Monitor Heart Rate and Blood Pressure: Regularly check your heart rate and blood pressure to ensure they stay within safe ranges.
Limit Cycle Length: Avoid prolonged cycles and take breaks to prevent tolerance buildup and reduce the risk of side effects.
Avoid Stimulants: Combining Clenbuterol with other stimulants (like caffeine) can amplify side effects, so limit other stimulant intake during use.
Consult a Healthcare Professional: Due to the potential risks, it’s best to consult a healthcare provider before starting Clenbuterol to understand the full implications.
Legal Status and Availability
Clenbuterol’s legality varies by country. In the United States, Clenbuterol is not approved for human use and is classified as a banned substance in many sports. However, it is still legal for veterinary use in some areas. In countries where Clenbuterol is legal for human consumption, it is typically available only by prescription.
Many individuals obtain Clenbuterol from online suppliers, but it’s crucial to ensure the product is genuine and sourced from reputable sellers, as counterfeit products can be dangerous.
Clenbuterol offers significant benefits for those looking to burn fat, retain muscle, and improve workout performance. However, it’s not without risks, and the potential side effects make it unsuitable for everyone. If you’re considering Clenbuterol, it’s vital to weigh the benefits against the possible health risks and to approach the drug with caution.
Conclusion
Clenbuterol can be an effective tool for those looking to accelerate fat loss, enhance muscle retention, and improve overall body composition. However, it is essential to use it responsibly, adhere to proper dosing guidelines, and monitor for side effects. Clenbuterol is not a magic solution but can be an effective supplement when combined with a solid diet, training program, and a safe approach.
0 notes
Text
UV Stabilizers Market- Industry Analysis, Market Size, Share, Trends, Application Analysis, Growth And Forecast 2024–2030.
UV Stabilizers Market Overview :
UV Stabilize Market size is forecast to reach $2,243.13 million by 2030, after growing at a CAGR of 6.4% during 2024–2030, owing to the wide usage of UV stabilizer in various applications such as furniture, architectural, automotive, packaging, and more. UV stabilizers filters harmful UV light and helps prevent colour change and delamination of coatings, adhesives, and sealants owing to which it is extensively used in these applications. The growing need for packaging and automotive due to rapid urbanization and increasing population in various regions, is the major factor driving the growth of UV stabilizers market during the forecast period. In addition, the growing demand of greenhouse films globally is anticipated to play a key role in driving the UV stabilizers market during the forecast period.

Market snapshot
UV Stabilizers Market Report Coverage
The report: “UV Stabilizers Market — Forecast (2024–2030)”, by IndustryARC, covers an in-depth analysis of the following segments of the UV stabilizers Industry. By Type: Hindered Amine Light Stabilizers, UV Absorbers, and Quenchers By Application: Coatings (Waterborne Coatings, UV Curable Coatings, and Others), Adhesives and Sealants, Plastics, and Others. By End-Use Industry: Architectural (Residential, and Commercial), Agriculture, Automotive (Automotive Interior, and Automotive Exterior), Furniture (Outdoor Furniture, and Indoor Furniture), Packaging (Beverage Packaging, Food Packaging, and Toiletries & Cosmetics Packaging), and Others By Geography: North Americas, South America, Europe, Asia Pacific, RoW
Inquiry Before Buying
Key Takeaways
Asia-Pacific region is poised to dominate the UV stabilizer market due to significant growth in construction, packaging, and automotive sectors across countries like China, India, and South Korea.
Light stabilizer solutions in which UV absorbers and HALS (Hindered amine light stabilizers) are synergistically combined, provide high-performance solutions for automotive OEM refinish paints, which tends to increase the market growth.
Even in indoor diffuse light conditions, naturally pale wood tends to yellow and darken with time as a result of lignin photo-oxidation. So to avoid photo-oxidation the UV stabilizers are widely used in the indoor furniture.
Request Sample
UV Stabilizers Market Segment Analysis — By Type
The hindered amine light stabilizers (HALS) segment held the largest share in the UV stabilizer market in 2023, as it efficiently inhibits polymer degradation. HALS high efficiency and longevity are due to a cyclic process wherein the HALS are regenerated rather than consumed during the stabilization process. Hindered amine light stabilizers are very efficient stabilizers for polymers and especially polyolefins. They do not absorb UV radiation but act to inhibit degradation of the polymer. In addition, significant levels of stabilization are achieved at relatively low concentrations on using hindered amine light stabilizers due to which its demand is increasing from various end-use industries. All these factors are boosting the demand for hindered amine light stabilizers (HALS) during the forecast period.
UV Stabilizers Market Segment Analysis — By Application
The flooring and decking segment is poised to capture the largest share of the market. The surge in UV stabilizer usage for flooring and decking is primarily attributed to the rising trend of outdoor living spaces among consumers. As the demand for functional and appealing outdoor areas grows, decking and patios have gained significant popularity. Additionally, UV stabilizers play a critical role in protecting flooring and decking materials from premature aging and weathering. Revenue generated from flooring and decking is closely linked to the construction industry, making its development crucial for UV stabilizer demand. Consequently, these factors within the flooring and decking segment are expected to propel the growth of the UV stabilizers market in the forecast period.
UV Stabilizers Market Segment Analysis — By End Use Industry
UV stabilizers play a crucial role in the packaging industry, serving as key components in materials like plastics to uphold packaging integrity and visual appeal throughout storage and transit. A notable trend within this realm is the escalating requirement for UV-resistant packaging materials, notably in the food and beverage sector, to preserve product quality and extend shelf life. Manufacturers are actively engaged in crafting UV stabilizers customized for specific packaging needs to address the industry’s changing dynamics. Conversely, the automotive segment is poised for the most rapid growth in the foreseeable future. UV stabilizers are indispensable in safeguarding various vehicle components, including interior materials and coatings, from UV radiation. An observable trend in this domain is the adoption of UV-stabilized materials to uphold the appearance and endurance of automotive parts exposed to sunlight. As automotive designs increasingly integrate plastics, there’s a mounting demand for UV stabilizers to ensure these materials endure UV exposure, thereby enhancing vehicle durability and aesthetics. The automotive industry remains a pivotal force propelling the UV stabilizers market forward
UV Stabilizers Market Segment Analysis — By Geography
North America leads the UV stabilizers market revenue share due to the strong presence of established automakers, which extensively use these stabilizers in vehicles. The automotive sector’s projected expansion in manufacturing will drive up demand for ultraviolet stabilizers in the foreseeable future. Plastic serves as a prevalent packaging material across diverse sectors such as food & beverage and PET bottles. The U.S. Food and Drug Administration ensures the safety of food packaging materials, particularly polymers in contact with food products. Additionally, the Asia Pacific region is poised to experience a significant Compound Annual Growth Rate (CAGR) in the global UV stabilizers market. China’s packaging sector is witnessing growth driven by technological advancements and research efforts in food packaging, meeting demands for safer and cleaner packaging solutions. Urbanization in China has intensified the need for processed and packaged foods as cities lack agricultural resources, further boosting the demand for food packaging materials.
Schedule a Call
UV Stabilizers Market Challenges
Fluctuations of UV stabilizers costs for raw materials
The costs of raw materials essential for UV stabilizer production, such as aluminum chloride, benzene, sodium nitrite, and benzoyl chloride, experience regular fluctuations. These fluctuations directly influence the pricing of UV stabilizers, consequently impacting market growth. Presently, benzene is priced at USD 696.7 per metric ton, sodium nitrite at USD 40 per kilogram, and benzoyl chloride at USD 38 per 1000 grams.
Limited awareness and adoption
UV stabilizers pose challenges for manufacturers in North America. Some industries and end-users may lack understanding regarding the benefits and applications of UV stabilizers. Educating the market about the significance of UV protection and the advantages of utilizing UV stabilizers is essential. Manufacturers must focus on building awareness and illustrating the value proposition of UV stabilizers to facilitate their adoption in various industries.
UV Stabilizers Market Drivers
Increasing Level of UV Radiation and The Harmful Effect of UV Radiation
The increased use of refrigerator and AC, which emits ozone-layered gasses, has led significantly to an increasing UV radiation level. NASA has stated that in the last 30 years, the amount of UV radiation has raised dramatically causing degenerative effects on sunlight materials. The increased use of gas emission devices enhances the growth of the UV stabilizer market with increased UV radiation. People are becoming increasingly aware about the harmful effects of different materials such as plastics and wood. Polymer photo degradation occurs when a substance is exposed to UV radiation and the physical characteristics of the substance, such as colour, loss of material strength, elongation and tensile strength characteristics or surface cracking, are deteriorated. The manufacturers therefore apply UV light stabilizers to prevent damage and avoid loss of components that are exposed to UV radiation. Thus, the increasing level of UV radiation and the harmful effect of UV radiation act as a driver for the UV stabilizers market growth.
Buy Now
Escalating Construction Sector
Continuing infrastructure growth has enabled construction industry to expand. Alternative inexpensive raw materials like plastic and wood products are preferred by businesses or manufacturing. However, these materials can be quickly shaped into various forms, which contribute to substantial demand for flooring, shielding and fencing applications. Such materials with high exposure to the sun cause degradation and UV stabilizers have been applied as a coating substrate on such materials to minimize such efficiencies. The rapid growth of housing and construction industry spending has increased dramatically in the U.S. and Mexico, resulting in the economic development of the region of North America. Eco-friendly use, low VOC emissions and high content of UV stabilizers are favored for the use of wood coating. Because of rising demand and the large building and construction market in the region, the country exports almost 90 percent of the furniture from US manufactures. This is a major factor in the growth of UV stabilizers in North America, such as HALS and UV absorbers. The growing use of plastics and wood in the building industry will thus drive the growth of ultraviolet stabilizers to preserve their intensity.
Market Landscape
Technology launches, acquisitions, and R&D activities are key strategies adopted by players in the UV stabilizers market. In 2023, the market of UV stabilizers has been consolidated by the top eight players accounting for 60% of the share. Major players in the UV stabilizers market are BASF SE, Solvay S.A., Songwon Industrial Co., Ltd., Everlight Chemical Industrial Corp., ADEKA Corporation, Mayzo Inc., Clariant AG., Addivant, Lycus Ltd. LLC, and Valtris Specialty Chemicals.
Acquisitions/Technology Launches
In April 2023, Damen Shipyards, in collaboration with AkzoNobel and Philips, developed a fouling control solution using UV-C light technology for ship hulls.
In March 2023, Clariant and Omya joined forces to address yellowing concerns in SMP sealants with the introduction of AddWorks IBC 760.
In July 2022, Solvay introduced a novel range of UV-C stabilizers tailored for hygiene applications, aimed at combating COVID-19 and reducing the incidence of hospital-acquired infections.
For more Chemicals and Materials Market reports, please click here
0 notes
Text
Propylene Oxide Market Dynamics: A Comprehensive Overview
Introduction Propylene oxide is an organic compound with the chemical formula C3H6O. It is a colorless volatile liquid that is miscible with water, most common organic solvents and has a faint ethereal odor. C3H6O is an epoxide, meaning it contains an oxide group bonded to two carbon atoms forming a three-membered cyclic ether. It is formed through the oxidation of propylene. Production of Propylene Oxide Industrially, propylene oxide is produced on a large scale through the chlorohydrin process or hydroperoxide process. In the chlorohydrin process, propylene is oxidized with chlorine in the presence of a chloride catalyst to form propylene chlorohydrin which is then hydrolyzed to yield C3H6O and hydrochloric acid. This process allows for greater selectivity in product formation. The hydroperoxide process involves three steps - first hydroperoxide is formed by oxidizing propylene with an oxygen containing gas like air or oxygen over a silver catalyst. This is followed by decomposition of hydroperoxide to form C3H6O and water. The final step recovers and purifies the C3H6O product. This process affords higher atom efficiency and lower energy consumption. Properties and Applications Due to its versatile functionality of forming additional bonds through ring opening, C3H6O has a wide range of applications. It is mainly used for producing polyether polyols which are building blocks for polyurethane products like rigid and flexible foams for insulation and cushioning. C3H6O is also used to produce propylene glycols that are commonly used as heat transfer fluids and manufacturing solvents. As an intermediate chemical, C3H6O is converted to propylene glycol ethers which are used in industrial and automotive antifreeze formulations as well as paints, coatings and cleaning agents due to their good solvent properties. Epoxy resins formed from reacting C3H6O with epichlorohydrin are used to produce protective and durable coatings, adhesives, composites and encapsulation of electronics. C3H6O acts as a intermediate in producing other chemical compounds like propylene glycol monomethyl ether and propylene glycol monomethyl ether acetate which are solvents for inks, coatings and electronics packaging. It can also react with amines to form amine oxide surfactants that are used in personal care, home care and industrial cleaning products. Health and Environmental Effects While C3H6O is useful industrially, it also poses certain health risks. It is a volatile flammable liquid and contact with eyes and skin can cause irritation. Inhalation may cause respiratory issues. Studies have shown it to be possibly carcinogenic to humans and its vapors are harmful if inhaled in large quantities. Organizations like the EPA have classified C3H6O as a hazardous air pollutant and possible human carcinogen. Manufacturing sites emitting this chemical are subject to regulations regarding its release into the environment. Proper handling and storage in a well-ventilated area along with use of protective equipment is important when working with propylene oxide. Spills contaminating soil and water need remediation to minimize environmental damage. Conclusion In summary, C3H6O is a versatile chemical building block found in many industrial and commercial products. While serving important applications, its adverse health effects require careful production and usage practices, transportation, storage and disposal according to safety guidelines. Ongoing research explores safer production technologies for this valuable oxirane compound supporting many modern industries. With proper risk management measures, propylene oxide will continue meeting manufacturing needs sustainably.
0 notes
Text
Test Bank For Organic Chemistry Eighth Edition Peter Vollhardt

Table of Contents
1. Structure and Bonding in Organic Molecules 2. Structure and Reactivity: Acids and Bases, Polar and Nonpolar Molecules 3. Reactions of Alkanes: Bond-Dissociation Energies, Radical Halogenation, and Relative Reactivity 4. Cycloalkanes 5. Stereoisomers 6. Properties and Reactions of Haloalkanes: Bimolecular Nucleophilic Substitution 7. Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 8. Hydroxy of Functional Group: Alcohols: Properties, Preparation, and Strategy of Synthesis 9. Further Reactions of Alcohols and the Chemistry of Ethers 10.Using Nuclear Magnetic Resonance Spectroscopy to Deduce Structure 11. Alkenes: Infrared Spectroscopy and Mass Spectrometry 12. Reactions to Alkenes 13. Alkynes: The Carbon 14. Delocalized Pi Systems: Investigation by Ultraviolet and Visible Spectroscopy Interlude 15. Benzene and Aromaticity: Electrophilic Aromatic Substitution 16. Electrophilic Attack on Derivatives of Benzene: Substituents Control Regioselectivity 17. Aldehydes and Ketones: The Carbonyl Group 18. Enols, Enolates, and the Aldol Condensation: a,b-Unsaturated Aldehydes and Ketones 19. Carboxylic Acids 20. Carboxylic Acid Derivatives 21. Amines and Their Derivatives: Functional Groups Containing Nitrogen 22. Chemistry of the Benzene Substituents: Alkylbenzenes, Phenols, and Benzenamines 23. Ester Enolates and the Claisen Condensation: Synthesis of b-Dicarbonyl Compounds; Acyl Anion Equivalents 24. Carbohydrates: Polyfunctional Compounds in Nature 25. Heterocycles: Heteroatoms in Cyclic Organic Compounds 26. Amino Acids, Peptides, Proteins, and Nucleic Acids: Nitrogen-Containing Polymers in Nature Read the full article
0 notes
Quote
Fast-scan cyclic voltammetry (FSCV) is cyclic voltammetry with a very high scan rate (up to 1×106 V·s−1).[1] Application of high scan rate allows rapid acquisition of a voltammogram within several milliseconds and ensures high temporal resolution of this electroanalytical technique. An acquisition rate of 10 Hz is routinely employed. FSCV in combination with carbon-fiber microelectrodes became a very popular method for detection of neurotransmitters, hormones and metabolites in biological systems.[2] Initially, FSCV was successfully used for detection of electrochemically active biogenic amines release in chromaffin cells (adrenaline and noradrenaline), brain slices (5-HT, dopamine, norepinephrine) and in vivo in anesthetized or awake and behaving animals (dopamine). Further refinements of the method have enabled detection of 5-HT, HA, norepinephrine, adenosine, oxygen, pH changes in vivo in rats and mice as well as measurement of dopamine and serotonin concentration in fruit flies.
Fast-scan cyclic voltammetry - Wikipedia
0 notes
Text
Meet Dactinomycin, aka Actinomycin D (get it? goofy ass name tbh). This very funky lil guy is a natural product found in soil and marine dwelling Streptomyces bacteria and has been used as an effective anti-cancer agent for a long time (as far back as 1954!).

The structure of dactinomycin immediately jumps out as particularly interesting. It’s composed of two cyclic peptides that are joined by a conjugated 3 ring system. The conjugated 3 ring system is actually also a chromophore (which means it absorbs light in the visible spectrum) and as a result this lil guy is actually a lovely red colour. The chromophore and bicyclic system are fairly unusual as far as peptides go but the way they come together to confer Dactinomycin’s cytotoxity is so cool.
Before I go on, there is still debate in the literature over dactinomycin’s exact mechanism (or mechanisms) of action, but the main one appears to be interference with the DNA to prevent transcription. The planar 3 ring system acts as an intercalator, meaning it inserts between the bases of double helical DNA (which is crazy, a peptide with an intercalation motif, so cool). Dactinomycin even specifically selects for two consecutive GC pairs, because the amine group on G is essential for forming a hydrogen bond with the lactones during intercalation. Some of the earlier studies on this are pretty neat, they investigated whether Dactinomycin intercalates by mixing DNA and Dactinomycin and then observing an increased viscosity of the DNA, which I think is an intuitive but nonobvious way to do it.
But that isn’t the whole story. The two peptide rings are thought to further stabilise the binding by forming additional hydrogen bonds with the phosphate groups in the DNA backbone. Crystal structures show the two ring systems sitting above and below the intercalation site, in a mirror image like arrangement as shown in this rather retro but still impressive image. They are even thought to confer additional specificity for poly GC regions of DNA.

There’s plenty more I could go into, like Dactinomycin’s remarkable membrane permeability and its clinical usage but this is already long and complex enough. Overall, Dactinomycin is a very useful peptide product with an unusual bicyclic structure and chromophore that jumped out at me as a pretty neat little molecule.
References
Liu, M. et al. (2019) ‘Identification of the actinomycin D biosynthetic pathway from marine-derived streptomyces costaricanus SCSIO ZS0073’, Marine Drugs, 17(4), p. 240. doi:10.3390/md17040240.
Dactinomycin (no date) National Center for Biotechnology Information. PubChem Compound Database. Available at: https://pubchem.ncbi.nlm.nih.gov/compound/Dactinomycin (Accessed: 22 May 2023).
Sobell, H.M. (1973) ‘The stereochemistry of actinomycin binding to DNA and its implications in molecular biology’, Progress in Nucleic Acid Research and Molecular Biology, pp. 153–190. doi:10.1016/s0079-6603(08)60103-8.
Meienhofer, J. and Atherton, E. (1973) ‘Structure—activity relationships in the actinomycins’, Advances in Applied Microbiology, pp. 203–300. doi:10.1016/s0065-2164(08)70028-5.
Images adapted from 1 and 3 respectively
1 note
·
View note
Text
im so ready to never take chem again
6 notes
·
View notes
Text
What is Polyphosphoric Acid and Why is it so important for Industrial Purpose?
Summary: Polyphosphoric acids are odorless, with high viscosity beverages with strong hygroscopic properties. Phosphoric acids containing less than 95% H3PO4 (68% P2O5) contain simple orthophosphoric acid.
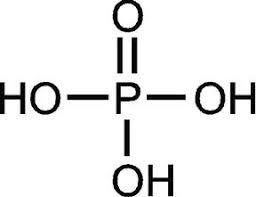
In high concentrations the acid contains a combination of ortho, pyro, tri, tetra and phosphoric acid which is highly concentrated. For this reason acids with a concentration greater than 68% P2O5 are better known as polyphosphoric acid. Polyphosphoric acid is insoluble in water, hydrolysing into orthophosphoric acid by heat production. They are insoluble in hydrocarbons and halogenated hydrocarbons. Polyphosphoric acid can be used as a reagent for cyclisation / ring in reactions such as acetylation, alkylation, amine production, acid amide, cyclic ketones, cyclic olefins or oxygen and heterocyclic sulfur compounds. Polyphosphoric acid can be used to make special support catalysts, e. g. used for the production of cumene from benzene. Polyphosphoric acid can be used to produce phosphate esters with a high mono-ester content. The acid is reacted directly with the alcohol necessary to produce the acidic phosphate ester.
Introduction:
Polyphosphoric Acid is made by diluting water and adding polymerizing to a technical or consuming phase of phosphoric acid, for example, in a full column or by absorbing P2O5 vapor into a recurrent stream of phosphoric acid. Polyphosphoric acid is used in many different applications, including the production of petroleum catalysts, surfactants for lotions and shampoos, oil extraction chemicals, industrial water treatment chemicals, flame structures, large cables and pigs, such as quinacridone pigments, and as additives. improved performance of petroleum products. Some industrial uses of polyphosphoric acid produce a phosphoric acid product that contains impurities, such as phosphate esters, other organic compounds and metals. Phosphoric acids can also be synthesized by other industrial processes, such as, for example, processes for the production of polycarbonate frames, synthetic fibers, pharmaceutical links and agricultural chemicals, and other industrial processes that use phosphorus-containing substances. These phosphoric acid products can be reduced and disposed of as a waste stream. Alternatively, those phosphoric acid products can be consumed by oxygen-by-product acids in the phosphoric acid production process, that is, the production of phosphoric acid by elemental phosphorus fire in excess air, thereby restoring phosphorus levels. by-product acid such as phosphoric acid and convert natural pollution into water and CO2.
Use of Polyphosphoric Acid:
Polyphosphoric acid can be used in the synthesis of aromatic sulfones, N-substituted amide and 4-aminobenzophenone. It can also be used for dehydration, Fischer-Indole Synthesis, Beckmann Rearrangement and Schmidt Rearrangement reaction. It can be used as a stimulant in the blending of various fragrant ketones. Phosphorus Oxychloride does not have a bright yellow, smoky, oily, and strong odor. It is used as a chlorinating agent, and in the manufacture of semiconductors, petroleum additives, plasticizers, hydraulic liquids, and organophosphorus compounds such as pesticides. PPA can be used as a composite during the synthesis of dimethyl carbonate (DMC) from urea and methanol. It also acts as an ammonia extract produced in this process. PPA can be used to prepare silica-based polyphosphoric acid, which is easy to carry, highly potent. Polyphosphoric acid (PPA) is widely used as an acylation and alkylation reagent in various reactions. A powerful mineral acid with excellent dehydration properties. It is made by diluting water and adding polymerizing to a technical level or by consuming phosphoric acid, for example, in a full column or by absorbing P 2O 5 vapor from the repeated distribution of phosphoric acid. Polyphosphoric acid can be used to make special support catalysts, e. g. used for the production of cumene from benzene. Polyphosphoric acid can be used to soften and lighten metal surfaces.
Conclusion:
Polyphosphoric acid can be used to make special support catalysts, e. g. used for the production of cumene from benzene. Polyphosphoric acid can be used to soften and lighten metal surfaces. Sandhya Group produces Polyphosphoric acid for industrial use.
3 notes
·
View notes
Text
Volte-Face: Chapter 2
Read on AO3 or FFN
Pairings: Christopher Pike x OFC (Aalin)
Rating: Teen
Word Count: 1k
Summary: On her first mission as a member of Starfleet, interpreter Aalin Matthews is stranded on a war-torn world ten days’ journey from a haven. Safe passage for Federation personnel has been revoked. Hostile forces are hunting the orphans she is protecting. As she and the children make the dangerous trek, the crews of both Enterprise and Shenzhou work behind the scenes to aid them.
Banned from intervening, from returning to the planet, from scanning for Aalin’s location, Captain PIke must decide whether to ignore the prohibitions or risk intensifying the conflict, a choice complicated by his personal feelings for Aalin. And Spock must come to terms with his decision allowing her to remain behind; should the worst occur, it will be the first time the young lieutenant has lost someone under his command. Both Pike and Spock undertake a dangerous mission among the brutal people who invaded their neighbors in order to protect Aalin and the children and end the war.
Excerpt: Ten Weeks Ago Enterprise
Commander Una called up a map of star system 3XAB9847 and addressed the assembled officers. “The early reports are true. Serenity was invaded by their nearest neighbor, the eighth planet in the system.”
“Serenity is our colloquial name for the planet,” Spock corrected. “In the indigenous language, it is known as Noohra, meaning light. This ancient term derives from the cyclic phenomena the original first contact team named Fireflies.” Enterprise’s science officer looked perplexed. “Which is an imprecise term. Actually it is a luminescent event in their eastern sky.”
“But Fireflies is so much more … I don’t know … romantic. It evokes the nostalgia of a warm summer’s night from childhood,” Trina Mann mused.
Spock raised an eyebrow, “Perhaps Ensign, but hardly accurate as no space insects are involved.” Boyce almost suppressed a chuckle. Spock continued without missing a beat, “Rather radiation is refracting off of ice flakes formed from the condensation of a comet trapped in high orbit by the planet’s gravity well.”
“See it’s already lost it’s magic,” Trina countered.
“All right guys let’s move on,” Pike intervened. “Number One, please continue.”
“It’s a typical scenario, Noohra is resource rich, with large deposits of a rare and valuable industrial mineral, primarily found in the mountainous region of the northern hemisphere. Aschask Prime is experiencing an industrial boom and its resources are nearly drained. About ten years ago, the Aschaski perfected the propulsion required to reach their nearest neighbor.”
Una continued, “Noohra’s population is clustered in the northern hemisphere, equally divided between cities near the oceans and small agricultural communities. Travel outside those cities is laborious and typically via a domesticated animal similar to a horse. Their technological capability is equivalent to the Asiatic region of Earth in the early 20th century prior to World War II.”
“Why was first contact made? It violates the Prime Directive.” Louvier, the Chief Engineer asked.
Pike answered, “It was accidental, a mistake made fifteen years ago which led to an encounter with the crew of the medical ship Jonas Salk. Since then we have walked a thin line trying to limit additional contamination of Noohra’s culture while repairing the damage from that meeting. Their central government has asked for humanitarian assistance from the Federation as well as help negotiating a cease-fire. Enterprise, along with the Shenzhou, will respond.”
“How will the Aschaski feel about that?” Amin, the navigator asked.
“Unknown,” Pike replied. “Phil, aid stations at the front lines and mobile hospitals in the hardest hit areas are our first priority. Any crewmember crossed trained as a medic is available to you.”
Una spoke next, “Ensign Mann, we will pick up supplies at Deep Space 5. Coordinate with the Shenzhou for their distribution planet-side. Amin, plot a speed run to the space station and then to Noohra. Spock, prepare a briefing on Aschask Prime for the bridge officers. Dismissed.”
Boyce, Louvier, the Security Chief, and Una remained. “And?” she asked.
“And what?” Pike asked, though he already knew the question.
“If the Aschaski blockade Noohra?”
“We help them, even if we have to shoot our way in,” their Captain answered.
Continue Reading on AO3 or FFN
Story Masterlist | Series Masterlist
OC Masterlist | Author Masterlist
#Christopher Pike#Christopher Pike x OC#Spock#Captain Pike#Captain Pike x OC#Captain Christopher Pike#Captain Christopher Pike x OC#Star Trek#Star Trek: Discovery#Star Trek: Strange New Worlds#christopher pike fanfiction#star trek fanfiction#fanfiction#fanfic#strange new worlds#Star Trek Discovery#Star Trek Strange New Worlds#star trek fanfic#christopher pike fanfic#star trek: aos#star trek aos
4 notes
·
View notes
Photo

Four Occasions and A Funeral
As above, so below… In layman’s terms, the impact of the celestial on the physical realm. It is an age old adage, upheld by our chosen souls. Yet the Unprecedented Romance proves that even a pervasive law, can be undermined by sheer will. Slowly, and then all at once, the cyclical nature of our souls are thrown off course. And what is left, but to pick up the pieces?
Our story begins with four occasions, and one funeral.
Occasion 1: A Loaded Invitation
The Rossi Family cordially invites you to pay homage to Ervig Rossi’s memory. Sunday, November 7th. 10AM at the Basillica di San Lorenzo. A service to follow immediately after at Palazzo Rossi.
On Monday, November 1st, seemingly innocuous invitations are hand-delivered by Janus’ souls to every involved party. With the Rossi family’s insignia and emblem, even the most daring souls cannot turn it away. Of course, it helps that each of Janus’ souls do so with the trademark gun holstered to their belt. The implication is in the messengers themselves - come, or be subject to suspicion. The messenger-and-recipient groups are as follows:
Lorenzo Rossi (Elder) - Ayesha Sharma, Valeria Greco, Edgar Ra
Zahra Njoki (Enterpriser) - Lee Ji-Hoon, Noelle Persaud, Wilhelmina D’Angelo
Andrea Rossi (Performer) - Yoo Ma-Ri, Akira Tanaka, Sevil Erdoğan
Javier Perez (Fighter) - Andy Alvarez, Courtney Goldmann, Julian Perez
Serena Madera (Fighter) - Miran Yıldız, Hayle Oliveira, Mercutio "Mercy" Kline
Sunghee Iseul (Healer) - Sloan Forbes, Safiya Amin, Valentine Garcia
Leo Blackwell (Healer) - Quinn Morelli, Sanem Çetin, Maximillian "Max" Madrigal
Cassandra Williams (Contender) - Bianca Howard, Bakhita Ahmadi, Santiago Herrera
Throughout the day, souls of Janus will visit their intended recipients with an invitation - and an implied ultimatum.
Occasion 2: Put it on the platinum card
There is no rest for the wicked - and business does not come to an end, simply because of loss and grief. As is tradition, the House of Mercury hosts a Happy Hour on Wednesday, November 3rd. Bank rolled by Mercury’s deep pockets, top-shelf liquor and exquisite cocktails are free to all patrons at the Grand Hotel Cavour’s exclusive bar. But there is no such thing as a free drink, as the souls of Mercury needle and prod any visitor. Where did their allegiances fall? What is the public sentiment around Ervig and Lorelai’s untimely deaths? It’s a question the Mercurian souls ask, as they sign away the bar tab with platinum cards.
Occasion 3: It’s their party - and they will cry if they want to
It has been a week and a half since Ervig Rossi and Lorelai Ann’s deaths. And though the House of Jupiter empathizes, the public’s need for catharsis cannot be ignored. Despite both Minerva and Janus’ impassioned disagreement, the show goes on in the form of The Amore’s doors opening. On the late hour of Friday, November 5th - The Amore reopens after a respectful period close. But the tantalizing reprieve only lasts for an hour, before an unexpected brawl between Minerva’s (Andy and Miran) and Janus’ (Serena and Javier) Fighters erupts on the dancefloor. The spectacle closes the doors on the event hours before last call - and one has to wonder if the fight is simply that, or a means to an end over the unpopular reopening.
The Funeral
It seems only appropriate that the House of Janus’ properties commemorate the loss of its fallen Elder. The week ends on Sunday November 7th, where Ervig Rossi’s tenure began - with a funeral service held at the Basillica di San Lorenzo. In the historic church, a sea of black attire occupies every pew. Is it sympathy or fear that draws a number of souls to the cathedral? The question is answered at Palazzo Rossi shortly thereafter, where many come to pay their respects, and kiss the ring of the new King - Lorenzo Rossi.
It is there that agendas come to light, with each House’s souls dedicated to a particular task:
Souls of Mercury in attendance are tasked with gaining favor with Janus’ souls. After all, everyone can profit in the aftermath of grief. Especially a house with a new fervor to rebuild.
Souls of Jupiter, emboldened by their Elder (Valeria), hold the insult of the fight at The Amore close to their chests. Their charismatic souls are tasked with assessing both grieving houses (Janus and Minerva) for weak spots - there is always a wayward soul, looking for a fresh start.
Souls of Minerva are tasked by their Elder (Edgar) to keep a steady resolve and fly under the radar. Quiet, observant, and respectful - even as the blood of their own, fallen member (Lorelain Ann) is still stained on their own hands.
Occasion 4: What is the way forward?
Not to be undone, the House of Minerva curtails the spectacle with one of their own. But unlike their rivals, who prove their agenda through intimidation, greed, and hedonism respectively, they lay in wait. Once the souls exit the Palazzo Rossi, Minerva’s souls in attendance are sent a simple text by Minerva’s Performer (Ji-Hoon) himself. Meeting at the Oblate Library at Midnight.
Under the midnight darkness, the souls of Minerva gather to their secret spot behind the Oblate Library’s archives. It is there that their Elder (Edgar) emerges from The Middle. In his expression, there is nothing but grief and darkness. His fists are tight, as he laments the God of Minerva’s message from The Middle.
“The souls of Lorelai and Ervig have vanished, but their impact remains. A perversion of this sort has consequences, and it is up to Minerva to serve it. The harbinger’s of justice must prevail, even if their means reflect the deadliest of sins.”
The repetition of Minerva’s words leave dumbfounded and fearful expressions on those in attendance. Until their Enterpriser, Yoo Ma-Ri speaks first. An assessment of Minerva’s words, that ripple in effect - “ruin, or be ruined.”
And just like that, the House of Minerva’s direction is set - destroy their rival houses, whatever means necessary.
--
Thank you to those who are ready to embark on the journey with us. If you have any questions, send us a message.
Next Event: A Holiday Season to Remember
4 notes
·
View notes
Text
Lupine Publishers|An Efficient Protocol for the One Pot Synthesis of Pyranopyrazoles in Aqueous Medium using Triethanolamine as a Catalyst

Abstract
Triethanolamine is an efficient and green catalyst for the synthesis of 6-amino-1, 4-dihydro-4-substituted-3-methylpyrano-[2, 3-c] pyrazole-5-carbonitrile in aqueous medium reflux conditions. The procedure is easier, eco friendly, simple with easy workup affording good yield of the corresponding products.
Keywords: Multi component reaction, Water media, Pyranopyrazole, Catalyst, Triethanolamine
Introduction
The present scenario for organic synthesis indicates the crave for green and economical synthesis of organic compounds. One of it is multi component synthesis. Strecker’s synthesis for amino acids was the first report on multi component reaction [1]. Last few decades show large development in it. The main aim of such reactions is to fasten the reaction rate by reducing number of steps involved and eventually increase the yield of reaction. In this context to achieve great efficiency catalysts are employed. Catalysts such as Nano α-Al2O3 supported ammonium dihydrogenphosphate [2], tungstate sulfuric acid [3], Fe3-xTixO4@SO3H nanoparticles [4], nano-titania sulfuric acid (15-nm TSA) [5], nanostructured MgO [6], H14[NAP5W30O110] [7] and ZnO Nanoparticles [8].
Organic catalysts such as Triethylamine [9] DABCO [10], Trishydroxymethyl aminomethane [11] are also reported in various organic transformations. Triethanolamine contains basic tertiary amine and primary alcoholic part (Figure 1).
It is used for activation of both CO2 and epoxides to convert them in to cyclic carbonates [12]. It is also reported as a legend for copper catalyzed hydroxylation of aryl halides in aqueous medium [13]. It is used as aqueous solvent for controllable preparation of ZnO nano flowers in sol gel technique [14]. Its aqueous solution is reported as electrolyte in CO2 Photo electro-conversion catalyzed by Cu- Doped Graphene-Titania Catalyst [15]. Also it is found to increase the rate of oxidation of mesitylene catalyzed by cobalt bromide [16]. It is used as sacrificial electron donor in photocatalytic system [17]. Furthermore; it improved the catalytic performance of CuBr/ PMDETA in the atom transfer radical polymerization [18]. It is also used as phase transfer catalyst for synthesis of 1-(arylsulfonyl) aryl/heterylmethanes [19]. It is used as medium for synthesis of 3-substituted coumarins using L-proline as a catalyst [20]. It is reported as catalyst in 10 mol% for synthesis of 2-amino-3-cyano- 4H-pyran derivatives under ultrasound irradiation at 600C [21].
Synthesis of substituted pyrano-[2,3-d]-pyrimidines via one-pot three-component condensation of aromatic aldehydes, malononitrile and barbituric acid or 2-thiobarbituric acid using trace amounts of ionic liquid (choline chloride.ZnCl2) and triethanolamine (0.1Mol%) at 75°C with stirring and under ultrasound irradiation [22] is also reported in literature. Herein we successfully attempted a fast and simple protocol for the synthesis of 6-amino-1,4-dihydro-4-substituted-3-methylpyrano [2,3-c]- pyrazole-5-carbonitrile by the one pot three component reaction of aromatic aldehyde, malononitrile and 3-methyl-1H-pyrazol-5(4H)- one using triethanolamine as a catalyst [23].
Results and Discussion
To explore the synthetic application of triethanolamine, in the present work we report the catalytic facet of it for the synthesis of heterocyclic compounds bearing pyrazole skeleton. To optimize the reaction conditions, we chose anisaldehyde as the prototype. Initially, 10mol% of triethanolamine was taken for solvent free reaction at room temperature. But the reaction afforded a low yield of the product after 2 hour stirring. Then we used 10ml of water for room temperature stirring [24]. After 2 hours stirring it gave 62% of yield. The yield of reaction gets drastically changed on increasing temperature. At 900C we got 85% of yield of the product. When 20mol% of triethanolamine was used then we got 92% of yield at 900C in 10 ml water. Other solvents were also studied expecting better yield but other than ethanol and water we got poor yields (Table 1). Further increase of temperature and amount of triethanolamine did not improve yield significantly (Table 1). After optimizing the reaction conditions, differently substituted aldehydes with electron donating as well as electron withdrawing groups were reacted to examine the feasibility of this catalytic reaction (Scheme 1).
Almost all aldehydes bearing various substituents such as –Cl, F, -NO2, -OMe etc afforded good yield of the corresponding products. All the synthesized compounds showed sharp peaks at 3410, 3356cm- 1(-NH2) and 2190cm-1(-CN) in IR spectra which supports for the formation of pyranopyrazole. The formed products being insoluble in water were easy to separate from the aqueous medium by simple filtration. The reason for catalytic activity of triethanolamine is it’s solubility in aqueous medium and basic nature. Products are simply purified by re crystallization with ethanol. Thus the protocol described herein is efficient for the synthesis of pyrazopyrazoles which do not need purification by column chromatography.
Model reaction* for anisaldehyde (2mmol), malononitrile (2mmol) and 3-methyl-1H-pyrazol-5(4H)-one (2mmol) using the above cited conditions @Isolated yield.
Experimental
Melting points were recorded in open capillaries and were uncorrected. Progress of reaction was monitored by TLC (30% of ethyl acetate: n-hexane). IR spectra were taken by KBr disc on Shimadzu IR Affinity 1 spectrophotometer [1]. H NMR spectra were recorded on a Varian 400MHz spectrophotometer in the specified solvents. Chemical shifts were expressed in 𝛿ppm relative to TMS. Mass spectra were recorded on a Macro mass spectrometer (Waters) by electro spray method (ES).
General method for the synthesis of 6-amino-1, 4-dihydro- 4-substituted-3-methylpyrano-[2,3-C]-pyrazole- 5-carbonitrile
To a stirred mixture of aromatic aldehyde (2mmol), malononitrile (2mmol) and triethanolamine (20mol %) in 10ml of water, 3-methyl-1H-pyrazol-5(4H)-one (2mmol) was added. The resulting mixture was stirred and heated at 900C for appropriate reaction time (Table 2). After completion of reaction, the reaction mixture was cooled, filtered off the residue as the crude product which was further purified by re crystallization form ethanol (Scheme 2).
Representative Spectral Data
6-Amino-1,4-dihydro-4-(4-methoxyphenyl)-3-methylpyrano[ 2,3-c]pyrazole-5-carbonitrile (4a)
White solid, [1]H NMR (400 MHz, DMSO-d6): 𝛿 ppm 12.08 (s, 1H), 6.87-7.23 (m, 4H), 6.81 (bs, 2H), 4.45 (s, 1H), 3.78 (s, 3H), 1.81 (s, 3H); IR (KBr) cm-1: 3425, 3128, 2928, 2200, 1597, 1153, 1203; ES-MS m/z: 283.2 (M+1)+.
6-Amino-2,4-dihydro-3-methyl-4-phenylpyrano[2,3-c] pyrazole- 5-carbonitrile (4e)
White solid, M.P. 245-246 0C; 1H NMR (400 MHz, DMSO-d6) : 𝛿 ppm 12.10 (s, 1H), 7.10-7.40 (m, 5H), 6.85 (s, bs, 2H), 4.60 (s, 1H), 1.78 (s, 3H); IR (KBr) cm−1 : 3410, 3356, 3167, 2990, 1646, 1596, 1399, 1276, 870; ES-MS m/z: 253 (M + 1) +.
6 - A m i n o - 4 - ( 4 - c h l o r o p h e n y l ) - 3 - m e t h y l - 2 , 4 - dihydropyrano[2,3-c]pyrazole-5-carbonitrile (4g)
Off-white solid, M.P. 230-2320C; 1H NMR (400 MHz, DMSO-d6): 𝛿 ppm 12.15 (s, 1H), 7.10–7.40 (m, 4H), 6.95 (s, bs, 2H), 4.63 (s, 1H), 1.80 (s, 3H); IR (KBr) cm−1 : 3478, 3035, 2985, 2193, 1647, 1596, 1398, 1284, 870; ES-MS m/z: 287 (M + 1) +.
6-Amino-4-(4-N, N-dimethylaminophenyl)-3-methyl-2, 4-dihydropyrano[2, 3-c]pyrazole-5-carbonitrile (4j)
Yellow solid, M.P. 234-235 0C; 1H NMR (400 MHz, DMSO-d6): 𝛿 ppm 12.10 (s, 1H), 6.70-7.15 (m, 4H); 6.55 (s, bs, 2H), 4.40 (s, 1H); 2.85 (s, 6H), 1.78 (s, 3H); IR (KBr) cm−1 : 3385, 3172, 2957, 2189, 1644, 1601, 1397, 1279, 868; ES-MS m/z: 296 (M + 1) +.
Conclusion
In summary, we have developed an efficient protocol for the synthesis of pyranopyrazoles by a simple method using a catalytic amount of triethanol amine. Herein; not only the yield of reaction is improved but also the reaction time is reduced. The workup of the reaction is very simple which make it easier to isolate the product.
#For more information#https://lupinepublishers.com/chemistry-journal/archive.php#Please click here#https://lupinepublishers.com/chemistry-journal/#Lupine Publishers#archive of organic and inorganic chemical sciences#Pyranopyrazole
5 notes
·
View notes
Text
Synthesis and Characterization of New Triazinane-2-Thione and Oxadiazinane 4-Thiones from Benzimidazole-Juniper Publishers
JUNIPER PUBLISHERS-OPEN ACCESS JOURNAL OF DRUG DESIGNING & DEVELOPMENT

Abstract
The present study a new strategy for the synthesis of most potent nitrogen heterocycles i.e., triazinane-2-thiones and oxadiazinanes-4- thiones (3-4) were efficiently synthesized by cyclic condensation based on classical Mannich amino methylation of N, N' unsymmetrical Thio ureas. The synthesized compounds have been characterized by analytical and spectral (IR, 1HNMR and Mass) data.
Keywords: Synthesis; Characterization; Triazinane-2-thione; Benzimidazole
Introduction
Demand for higher efficiency, economy and selectivity in the synthesis of novel molecular scaffolds drives organic chemistry, particularly nitrogen-containing molecules has been of longstanding interest to organic chemists due to their great importance in chemistry and biology [1]. Many of these heterocyclic compounds exhibit various biological activities [2,5]. Little work has been published in the area of 1,3,5 triazinane- 2-phones showing antimicrobial activity [6], Biocidal [7] and enantio-differentiating coupling reagents [8]. The potent Heterocycles having, structural unit has a significant place among pharmaceutically important synthetic and natural materials [9].
Materials and Methods
Melting points of the synthesized compounds are determined in open capillary tubes and are uncorrected. Reaction Progress was observed by TLC plates, Bruker 300MHz instrument was used to record 1HNMR spectra in DMSO/CDCl3 using TMS as internal standard. Chemical shifts (5) are expressed in ppm. Perkin Elmer BX series FT-IR was to record IR spectra, Elemental analysis were performed on a PerkinElmer 240CHN analyzer Figure 1.
Experimental section
Synthesis of 1-(5-(4-(1H-benzo[d]imidazol-2-yl)phenyl) amino)thiazol-2-yl)-3-phenyl thiourea (2): A mixture of compound [10] (1) (0.01mmol) and sodium hydride (0.5g, 20mmol) in Ethanol (80ml) was heated under reflux for 30min and cooled. Phenyl isothiocynate (0.01mmol) was added and refluxing continued for a further 4h. The solvent was evaporated off and the residue dissolved in DCM (50ml) and washed with dilute HCl. The organic phase was dried (MgSO4) and the solvent evaporated off to give the desired compound (2). The progress of the reaction was monitored by TLC and recrystallized from ethanol.
Synthesis of 1-(5-(4-(1H-benzo[d]imidazol-2-yl) Phenylamino)thiazol-2-yl)-5-methyl-3-phenyl-1,3,5 triazinane-2-thione (3): A mixture of compound (2) in (1mmol), 30% formaldehyde (2mmol) and methyl amine (1mmol) and (0.01mole) NaOH were taken in ethanol (30ml) and refluxed for about 5-6h. The progress of the reaction was monitored by TLC. After completion of the reaction it was cooled and the product was filtered. The crude product was passed through silica gel by column and the product was eluted from 60% ethyl acetate and hexane.
Synthesis of 3-(5-((4-(1H-benzo[d]imidazol-2-yl) phenyl)amino)thiazol-2-yl)-5-phenyl-1,3,5-oxadiazmane- 4-thione(4): A mixture of compound (2) in (1mmol), 30% formaldehyde (2mmol) were taken in ethanol (30ml) and add 1ml concentrated HCl (5ml) and refluxed for about 5-6hr at 100110 °C. The progress of the reaction was monitored by TLC. After completion of the reaction it was cooled and neutralized with 10% NaOH. The product was filtered. The crude product was passed through silica gel by column and the product was eluted from 60% ethyl acetate and hexane. These compounds were purified by recrystallisation from suitable solvents. Structures of the synthesized compounds [2-4(a-e] have been confirmed on the basis of analytical and spectral (IR, 1HNMR and Mass) data (Table 1).
Results and Discussion
In the present study, it has been discussed that the synthesis of various benzimidazole fused triazinane 2-thione heterocyclic moieties by cyclic condensation based on classical Mannich amino methylation of N,N' unsymmetrical this ureas (2) with 30% HCHO and methyl amine in ethanol. An aliphatic amine has been yielded in the condensation between methyl amine and formaldehyde, it cyclizes immediately to produce corresponding 1,3,5-triazinane since imine is unstable. With respect to biological activity benzimidazole fused heterocyclic compounds such as triazinane derivatives are of very important than the cyclic compounds [11,12] (Table 2).
Conclusion
The research study reported efficiently synthesized new analogs of triazinane and oxadiazinane by classical amino methylation with different aerial-N,N' unsymmetrical thioureas by taking benzimidazole as starting material. All the compounds were characterized by standard spectroscopic techniques.
Acknowledgement
The authors are grateful acknowledge to the Department of chemistry Kakatiya university, Warangal for their constant support during this research work.
For more Open Access Journals in Juniper Publishers please click on: https://www.crunchbase.com/organization/juniper-publishers
For more articles in Open Access Novel Approaches in Drug Designing & Development please click on: https://juniperpublishers.com/napdd/index.php
For more Open Access Journals please click on: https://juniperpublishers.com
To know more about Juniper Publishers please click on: https://juniperpublishers.business.site/
1 note
·
View note