#southern Peru
Text

Machu Picchu is an Incan citadel set high in the Andes Mountains in Peru, above the Urubamba River valley. Built in the 15th century and later abandoned, it’s renowned for its sophisticated dry-stone walls that fuse huge blocks without the use of mortar, intriguing buildings that play on astronomical alignments and panoramic views. Its exact former use remains a mystery. Wikipedia
24 notes
·
View notes
Photo

Credit: Cristhian Hernandez
#Peru#Skull#Horns#Cattle#bovine#western#longhorn#cow#rural#rancher#farmer#farmland#homestead#ranch#bull#southern#farm#countryside#cowboy#cowgirl#agriculture#pasture#dairy farm#rodeo#barn yard#farm yard#cow skull
288 notes
·
View notes
Text

#Argentina#Paraguay#the Philippines#Southern Brazil#Southern France#Spain#Uruguay#Peru#Ecuador#Colombia#Chile#polls#alfajor
258 notes
·
View notes
Photo

Southern Viscacha (Lagidium viscacia)
by Oscar Fuentes
#southern viscacha#viscacha#rodents#Lagidium viscacia#lagidium#Chinchillidae#rodentia#mammalia#chordata#wildlife: peru#wildlife: south america
109 notes
·
View notes
Text
First Set of Matches coming soon, listed below so you can begin working on your propaganda
1. England vs Peru
2. Conch Republic vs Botswana
3. Liberia vs Guinea-Bissau
4. Kingdom of Talossa vs Johnston Atoll
5. Principality of Seborga vs Northern Cyprus
6. Montenegro vs Sao Tome and Principe 7. East Timor vs Saint Helena Ascension and Tristan da Cunha
8. Croatia vs United States Virgin Islands
9. Hajduk Republic of Mijat Tomic vs French Southern and Antarctic Lands
10. Kingdom of Wallachia vs Gabon
#nations of the world tournament#Propaganda encourages#England#Peru#Conch Republic#Botswana#Liberia#Guinea-Bissau#Kingdom of talossa#Johnston atoll#Principality of Seborga#Northern Cyprus#Montenegro#sao tome and principe#East timor#Saint Helena Ascension and Tristan da Cunha#Croatia#United States Virgin Islands#Hajduk Republic of Mijat Tomic#French Southern and Antarctic Lands#Kingdom of Wallachia#Gabon
1 note
·
View note
Video
youtube
Peru's Southern Copper Starts Legal Action To Evict Illegal Miners #per...
#youtube#Peru's Southern Copper Starts Legal Action To Evict Illegal Miners. peru americas latin copper mining apurimac mexico Southern Copper one
0 notes
Text
Prophet Muhammed was from the Americas
Prophet Muhammed was from the Americas is a necessary post to demonstrate that Maurs/ Mu’urs and Islam are indigenous to the Americas since people want to make Islam, via, the double O European Moors, as the invaders, when we know that the Dum Diversas (papal bull) of 1452 authorized Christians to take possession of Saracens (Moslem Maurs) and pagans (Hebrews) and subject them to perpetual…

View On WordPress
#Al Takwa#Hajj to Mecca#History of Islam#Holy City of Mecca#Islam in America#Kaba#Kabah#Koba#Medina#Moors#Naga-Maya#original Arabs#Origins of Islam#Prophet Muhammed#Southern Arabia#the Garden of Eden#The Kaaba#the original compass#Waka El Peru#Wakanda#Yucatan Peninsula
0 notes
Text


Rio Santiago Poison Frog (Excidobates captivus), family Dendrobatidae, found in NW Peru and southern Ecuador
photographs by Aiosha Estrada
600 notes
·
View notes
Photo








Shag carpet caterpillar moth, Prothysana felderi, Apatelodinae (American silkworm moths)
Found from very southern Mexico down through Peru
Photo 1 by Andreas Kay, 2 by Judy Gallagher, 3-4 by kkrockytop, 5 by desertnaturalist, 6 by indianacristo, 7 by erlandreflingnielsen, and 8 by kkrockytop
#animals#curators on tumblr#insects#bugs#moth#caterpillar#larva#american silkworm moth#Apatelodinae#shag carpet caterpillar#shag carpet caterpillar moth#one nice bug
670 notes
·
View notes
Text
Hiking a trans pride flag up 131 mountains in the Northern Appalachian/Adirondack Regions, parts 25-29: Southern Vermont and Western Mass!!!
Greylock - 29/131 (tallest mountain in Massachusetts):

Peru - 26/131:

Berlin - 28/131:

Stratton - 27/131:

Styles - 26a/131 (POV: you just said something transphobic on the mountain):

Dorset - 25/131:

Dorset Firetower - 25a/131 (I look so out of it in this one lmao):

More nature photos!!!



Thanks for reading this far :)
#lol flag time#pride#pride flag#trans#trans pride#transgender#transgender pride#queer#queer pride#lgbtq#lgbtq pride#lgbt pride#hiking#peakbagging#nature
1K notes
·
View notes
Text







Juan Borja, Spiriti della Foresta (Forest Spirits). Upcoming exhibition 12th of september – 31st of october 2023, Fcf Gallery, Milano.
Juan is a photographer and environmentalist from Peru. "My inspiration comes from photographers like the Vargas brothers and Martín Chambi in the southern andes, the mexicans Graciela Iturbide, Juan Rulfo and Gabriel Figueroa, Felice Beato in Japan, the brazilian Sebastiao Salgado and Karl Blossfeldt from Germany, as well as pictorialists like Misonne."
2K notes
·
View notes
Text
For #Caturday:

Ceramic bottle modeled in the form of a standing feline, decorated with resist-painted motif.
Gallinazo style (aka Virú culture), NW Peru, Early Intermediate Period, c. 200 BCE - 600 CE.
Spotted at the American Museum of Natural History NYC.
PS: this vessel may depict the Peruvian subspecies of Pampas Cat aka Northern Colocolo (Leopardus colocola garleppi). The Andean Mountain Cat (Leopardus jacobita) is also often suggested, but their range is more southern and higher elevation than where the Virú were? Also note the stripier legs on the Colocolo similar to the ceramic:
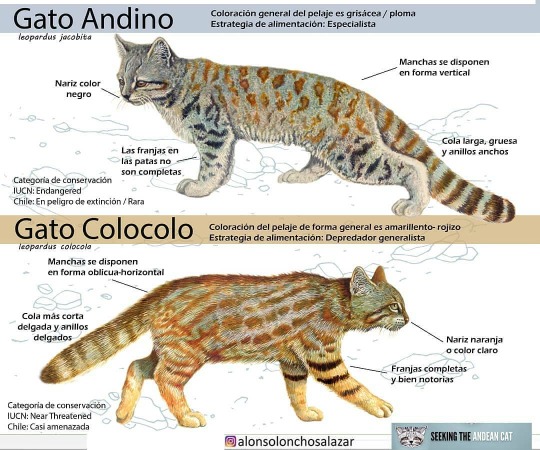
#cat#cats in art#feline#ceramics#effigy vessel#pre-conquest#Peruvian att#South American art#Indigenous art#Gallinazo#Virú#American Museum of Natural History#museum visit#Caturday#animals in art#Pampas Cat#Colocolo#Andean Mountain Cat#wild cats#mammalogy#zoology#species ID#ID#infographic#Peruvian art
3K notes
·
View notes
Text

Machu Picchu, Peru: Machu Picchu is a 15th-century Inca citadel located in the Eastern Cordillera of southern Peru on a 2,430-meter mountain ridge. Often referred to as the "Lost City of the Incas", it is the most familiar icon of the Inca Empire. It is located in the Machupicchu District within Urubamba Province above the Sacred Valley, which is 80 kilometers (50 mi) northwest of Cusco. Wikipedia
327 notes
·
View notes
Text
Fish of the Day
Today's fish of the day is the bonnethead shark!

The bonnethead shark, also called bonnet shark, or shovelhead, scientific name Sphyrna tiburo, is known for its unique head shape. It is often thought that the bonnethead shark is a young hammerhead shark, and despite belonging to the same family, Sphyrnidae, they are not the same species. Unlike the hammerheads worldwide range, the bonnethead shark's natural range only stretches from Southern Canada down to The coast of Brazil and Peru, with populations on both coasts of the Americas. It lives primarily in estuaries or bays, living around vegetation, sandy bottomed areas, or reefs. These are a migratory species: during the summer they move inshore and further North, but during Winter they move back south. Eastern populations concentrate around the Carolinas in Summer, and the Florida coast or Caribbean sea during the other months, but there are no concentrated points known for Pacific populations.

Bonnethead sharks, similar to other sharks of their size, eat primarily crustaceans. Crabs, shrimp, mollusks, and small fish. Similar to hammerhead sharks, they have many electromagnetic sensors on the underside of the head, this is the reason for the spade shape as the spacing of the sensors allows them to better find prey below them. They hunt along the sea bed, moving the head similar to how one moves a metal detector, looking for electromagnetic irregularities produced by living beings. After detection, the shark turns sharply and bites into the sediment, then grinding prey and swallowing. When unable to find prey, or in larger groups they also have been found eating seagrass. Other than for detection of prey, this head can also be used for better vision: as the eyes are faced to the sides this gives them a much wider field of view, allowing them to see if any predators are around, something that gives them a higher chance of surviving past childhood.

The reproduction of the bonnethead shark is viviparous, meaning that the baby is formed inside the mother and born alive. breeding is thought to take place around spring and autumn in the Atlantic populations, but may take place year round without a proper season for it. However, these fish will breed only after they've reached 31 inches in females, and 24 inches in males at around their second year of life. Then, after breeding, gestation takes only 5 months before they have anywhere between 4-12 pups, who are already 12 inches in length. These pups are then abandoned, primarily to keep the parents from feeding on their own offspring, and will live in and around seagrass beds they were born in. This is where they can hide from predators for the first few years. The largest bonnethead shark reported was 4 feet in length, but most are only 2.5-3 feet long. Male and female bonnetheads can be told apart by the sexual dimorphic morophology of the head shape. In females, the head is rounded, but males have a bulge shape around the midline of the head, referred to as a cephalofoil.

Their behavior is unlike many sharks, as bonnetheads are known to exist in groups. Living in smaller numbers of 5-15 in a pod, although there have been schools of thousands reported to be traveling with one another. These groups are not territorial, but they have a hierarchy within them. Strangest of all, these sharks appear to communicate with one another using cerebrospinal fluid as a chemical communicator between individual sharks, letting the others know where they are at any given time.

That's the bonnethead shark, everybody! Have a wonderful day.
#fish#fish of the day#fishblr#fishposting#aquatic biology#marine biology#freshwater#freshwater fish#animal facts#animal#animals#fishes#informative#education#aquatic#aquatic life#nature#river#ocean#shark#sharks#bonnethead#bonnethead shark#bonnet shark#shovel shark#shovelhead shark#hammerhead shark
124 notes
·
View notes
Text

May 31, 2024 - Jelski's Chat-Tyrant (Silvicultrix jelskii)
Found in the Andes in western Peru and southern Ecuador, these tyrant flycatchers live at the edges of mountain forests, often near streams. Usually foraging alone and occasionally joining mixed-species flocks, they eat insects, capturing prey in short flights from low perches or sometimes from the ground. Nothing is known about their breeding behavior.
#jelski's chat-tyrant#tyrant flycatcher#silvicultrix jelskii#bird#birds#illustration#art#water#birblr art
93 notes
·
View notes
Text


The archeocete Perucetus colossus dives through a coastal bloom of jellyfish in the Pisco Basin (southern Peru), some time during the Eocene (with bonus multiview).
I originally intended to add epibionts to this reconstruction (reflecting the specialized communities found on many living whales, especially baleen whales). Yet, interestingly, it appears that most animal epibionts and ectoparasites of modern cetaceans, such as whale barnacles (Hayashi et al. 2013) and remoras (Friedman et al. 2013), only appeared in the Neogene or late Paleogene, or have a poorly known (co-)evolutionary history, like whale lice (Pfeiffer 2009, Iwasa-Arai & Serejo 2018) and pennellids (large parasitic copepods) (Hermosilla et al. 2015). So, no epibionts* for big lad Perucetus!
References and notes about the reconstruction:
*animal epibionts. Unicellular eukaryotes like diatoms were most likely present on early cetaceans, given their prevalence on modern large marine animals (Ashworth et al. 2022). Of course, it is possible that other animals (i.e., early, less specialized representatives of modern groups, or different taxa altogether) were also already exploiting the surfaces offered by these early whales; however, this remains entirely speculative.
The reconstruction of Perucetus proposed in its original description (Bianucci et al. 2023) includes some rather odd (if interesting) choices about soft tissues, including limbs with webbed and distinguishable fingers, and a manatee-like tail. While these choices might be defendable in light of the rather basal status of Perucetus among cetaceans, I opted for a more derived look based on the assumption that fully marine cetaceans like basilosaurids would have probably rapidly acquired hydrodynamically favorable adaptations, pushing them towards a more familiar Neoceti-like appearance (even though Perucetus itself was likely a poor swimmer (Bianucci et al. 2023), it seems likely to me that this was a secondarily acquired trait, given the less extreme morphology of other basilosaurids).
Reconstruction in the multiview scaled to ~18 m in length after the estimations of Bianucci et al. (2023).
References:
Ashworth, M. P., Majewska, R., Frankovich, T. A., Sullivan, M., Bosak, S., Filek, K., Van de Vijver, B., Arendt, M., Schwenter, J., Nel, R., Robinson, N. J., Gary, M. P., Theriot, E. C., Stacy, N. I., Lam, D. W., Perrault, J. R., Manire, C. A., & Manning, S. R. (2022). Cultivating epizoic diatoms provides insights into the evolution and ecology of both epibionts and hosts. Scientific Reports, 12(1), Article 1. https://doi.org/10.1038/s41598-022-19064-0
Bianucci, G., Lambert, O., Urbina, M., Merella, M., Collareta, A., Bennion, R., Salas-Gismondi, R., Benites-Palomino, A., Post, K., de Muizon, C., Bosio, G., Di Celma, C., Malinverno, E., Pierantoni, P. P., Villa, I. M., & Amson, E. (2023). A heavyweight early whale pushes the boundaries of vertebrate morphology. Nature, 620(7975), Article 7975. https://doi.org/10.1038/s41586-023-06381-1
Friedman, M., Johanson, Z., Harrington, R. C., Near, T. J., & Graham, M. R. (2013). An early fossil remora (Echeneoidea) reveals the evolutionary assembly of the adhesion disc. Proceedings of the Royal Society B: Biological Sciences, 280(1766), 20131200. https://doi.org/10.1098/rspb.2013.1200
Hayashi, R., Chan, B. K. K., Simon-Blecher, N., Watanabe, H., Guy-Haim, T., Yonezawa, T., Levy, Y., Shuto, T., & Achituv, Y. (2013). Phylogenetic position and evolutionary history of the turtle and whale barnacles (Cirripedia: Balanomorpha: Coronuloidea). Molecular Phylogenetics and Evolution, 67(1), 9–14. https://doi.org/10.1016/j.ympev.2012.12.018
Hermosilla, C., Silva, L. M. R., Prieto, R., Kleinertz, S., Taubert, A., & Silva, M. A. (2015). Endo- and ectoparasites of large whales (Cetartiodactyla: Balaenopteridae, Physeteridae): Overcoming difficulties in obtaining appropriate samples by non- and minimally-invasive methods. International Journal for Parasitology: Parasites and Wildlife, 4(3), 414–420. https://doi.org/10.1016/j.ijppaw.2015.11.002
Pfeiffer, C. J. (2009). Whale Lice. In W. F. Perrin, B. Würsig, & J. G. M. Thewissen (Eds.), Encyclopedia of Marine Mammals (Second Edition) (pp. 1220–1223). Academic Press. https://doi.org/10.1016/B978-0-12-373553-9.00279-0
#'a heavyweight early whale pushes the boundaries of...' blablabla you've all read it by now#i have nothing to add#it's fat#look at it#that is all#perucetus#cetacean#mammal#vertebrate#eocene#cenozoic#paleontology#palaeoblr#paleoart#my art
420 notes
·
View notes